

Executive Summary
Ionic bonds form when one atom transfers electrons to another atom, creating positively and negatively charged ions that attract each other through electrostatic forces. Covalent bonds form when two atoms share electrons equally or unequally to achieve stable electron configurations. The key difference: ionic bonds involve complete electron transfer between atoms with very different electronegativities (typically metal + nonmetal), while covalent bonds involve electron sharing between atoms with similar electronegativities (typically nonmetal + nonmetal).
Use the electronegativity difference to identify bond type: differences greater than 1.7 indicate ionic bonding, while differences less than 1.7 indicate covalent bonding. Ionic compounds typically have high melting points, conduct electricity when dissolved or molten, and form crystalline solids. Covalent compounds typically have lower melting points, do not conduct electricity, and can exist as gases, liquids, or solids at room temperature.
This guide explains both bond types with clear examples from materials science, helping you understand their properties and applications in advanced materials and technology.
Table of Contents
1. Quick Comparison: Ionic vs Covalent Bonds
Ionic bonding occurs between atoms with very different electronegativities - one atom strongly attracts electrons while the other readily loses them. The atom that loses electrons becomes a positively charged cation, and the atom that gains electrons becomes a negatively charged anion. These opposite charges create strong electrostatic attraction, forming an ionic bond.
Covalent bonding occurs between atoms with similar electronegativities that both need electrons to achieve stable configurations. Instead of one atom taking electrons from another, both atoms share electrons. This sharing allows both atoms to fill their outer electron shells simultaneously, creating a stable molecule.
The Fundamental Difference
Think of ionic bonding like a complete transaction: one atom gives up electrons entirely, and another atom receives them. Think of covalent bonding like a partnership: both atoms contribute electrons to a shared pool that benefits both partners equally or unequally depending on their relative electronegativities.
Decision Rule: Check the periodic table positions. Metal + nonmetal → ionic bond. Nonmetal + nonmetal → covalent bond. For precise identification, calculate electronegativity difference (explained in Section 4).
2. Ionic Bonds Explained
Definition
An ionic bond is the electrostatic attraction between oppositely charged ions formed through complete electron transfer from one atom to another. The atom that loses electrons becomes a positively charged cation (typically a metal), and the atom that gains electrons becomes a negatively charged anion (typically a nonmetal). The resulting ionic compound consists of a three-dimensional lattice structure where each ion is surrounded by ions of opposite charge.
How Ionic Bonds Form
Step 1: Electron Transfer
The metal atom (low electronegativity) has loosely held valence electrons in its outer shell. The nonmetal atom (high electronegativity) has a strong attraction for electrons and nearly full outer shell. When these atoms approach, the metal transfers one or more valence electrons to the nonmetal to achieve noble gas electron configurations for both atoms.
Example: Sodium (Na) has one valence electron in its outer shell. Chlorine (Cl) has seven valence electrons and needs one more to complete its outer shell. Sodium transfers its single valence electron to chlorine.
Step 2: Ion Formation
After electron transfer, both atoms become ions. The metal atom, having lost electrons, now has more protons than electrons and becomes a positively charged cation. The nonmetal atom, having gained electrons, now has more electrons than protons and becomes a negatively charged anion.
Example: Sodium loses one electron to become Na⁺ (11 protons, 10 electrons). Chlorine gains one electron to become Cl⁻ (17 protons, 18 electrons). Both ions now have stable electron configurations matching the nearest noble gases (neon for Na⁺, argon for Cl⁻).
Step 3: Electrostatic Attraction
Opposite charges attract according to Coulomb's law. The positively charged cation and negatively charged anion experience strong electrostatic attraction that pulls them together. In solid ionic compounds, millions of ions arrange in a regular, repeating three-dimensional crystal lattice where each cation is surrounded by anions and vice versa, maximizing attractive forces and minimizing repulsive forces.
Properties of Ionic Compounds
High Melting and Boiling Points: Strong electrostatic forces between ions require significant energy to overcome. Sodium chloride melts at 801°C. Magnesium oxide melts at 2,852°C due to stronger attraction between Mg²⁺ and O²⁻ compared to Na⁺ and Cl⁻.
Electrical Conductivity: Ionic solids do not conduct electricity because ions are locked in fixed positions in the crystal lattice. However, molten ionic compounds or ionic compounds dissolved in water conduct electricity because ions are free to move and carry charge.
Solubility in Water: Many ionic compounds dissolve in polar solvents like water. Water molecules surround individual ions (hydration), separating them from the crystal lattice and stabilizing them in solution. The negative oxygen end of water molecules surrounds cations, while the positive hydrogen end surrounds anions.
Brittle and Hard: Ionic crystals are hard because strong electrostatic forces resist deformation. However, they are brittle because shifting the crystal lattice even slightly brings like charges next to each other, creating repulsive forces that cause the crystal to fracture rather than bend.
Real-World Examples
Sodium Chloride (NaCl): Table salt. Na⁺ and Cl⁻ ions arrange in a cubic crystal lattice. Used in food preservation, chemical manufacturing, and water softening.
Magnesium Oxide (MgO): Refractory material in high-temperature furnaces and crucibles. The Mg²⁺ and O²⁻ ions create exceptionally strong ionic bonds, resulting in a melting point above 2,800°C.
Calcium Carbonate (CaCO₃): Limestone, marble, chalk. Ca²⁺ ions bond with polyatomic carbonate ions (CO₃²⁻). Used in construction, agriculture (soil pH adjustment), and calcium supplements.
Lithium Cobalt Oxide (LiCoO₂): Cathode material in lithium-ion batteries. Li⁺ ions can move between crystal layers during charging and discharging, enabling energy storage in smartphones, laptops, and electric vehicles.
3. Covalent Bonds Explained
Definition
A covalent bond is the sharing of one or more pairs of electrons between two atoms, typically nonmetals with similar electronegativities. Instead of complete electron transfer, both atoms contribute electrons to a shared electron cloud that exists in the space between the nuclei. This shared electron pair attracts both nuclei simultaneously, holding the atoms together and forming a molecule.
How Covalent Bonds Form
Step 1: Electron Sharing
When two nonmetal atoms approach each other, both atoms have partially filled outer electron shells and need additional electrons to achieve stable noble gas configurations. Rather than one atom taking electrons from the other (which would be difficult since both atoms have similar electron affinities), both atoms share electrons.
Example: Two hydrogen atoms each have one electron and need one more electron to achieve helium's stable configuration (two electrons). Each hydrogen contributes its single electron to form a shared pair that both atoms can "count" as part of their electron shells.
Step 2: Orbital Overlap
Atomic orbitals from each atom overlap in the region between the two nuclei. The shared electron pair occupies this overlapping region, creating a bonding molecular orbital. The electron density between the nuclei reduces repulsion between positively charged nuclei while creating attraction that holds the atoms together.
Example: In H₂, the 1s orbitals from each hydrogen atom overlap, creating a sigma (σ) bond - the strongest type of covalent bond formed by head-on orbital overlap along the internuclear axis.
Step 3: Molecule Formation
Once atoms share electrons to satisfy their electron requirements, a stable molecule forms. The number of shared electron pairs determines the bond order: single bond (one shared pair), double bond (two shared pairs), or triple bond (three shared pairs). Higher bond orders mean stronger, shorter bonds.
Example: Oxygen (O₂) forms a double bond by sharing two pairs of electrons. Nitrogen (N₂) forms a triple bond by sharing three pairs of electrons. Water (H₂O) forms two single bonds between oxygen and each hydrogen atom.
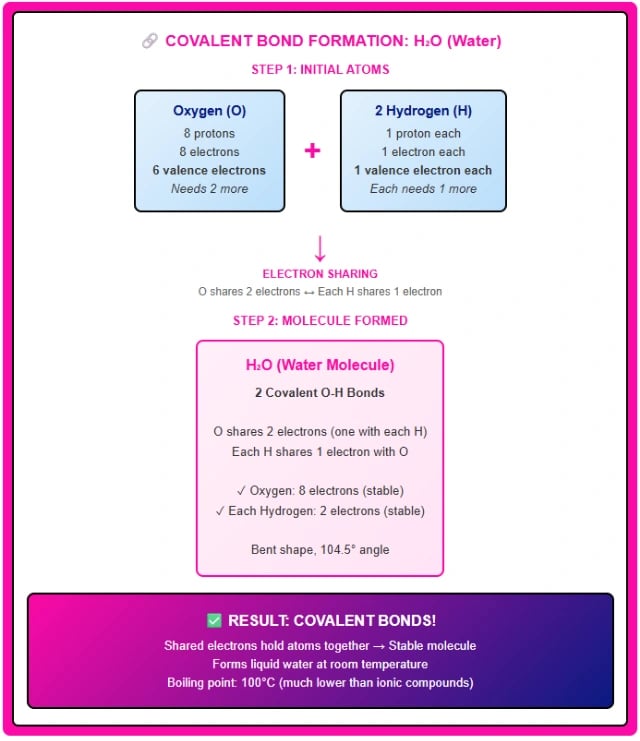
Step 1: Identify the elements involved.
Step 2: Are both elements nonmetals? YES → Covalent bond. NO → Continue.
Step 3: Is one element a metal and the other a nonmetal? YES → Ionic bond. NO → Metallic bond.
For precise identification with borderline cases (metalloids like silicon, boron), calculate electronegativity difference.
Related Articles
Continue exploring chemical bonding and materials science:
Stratified vs Cluster Sampling: Data Science Guide - Statistical methods for materials research
Voice Biomarkers for Cognitive Decline Detection - Bioengineering applications
Wearable Technology for Elderly Health Monitoring - Sensor materials and biocompatibility
Sources and References
Chemical Bonding - Wikipedia. Overview of ionic and covalent bonding mechanisms. Wikipedia article
Ionic Bonding - Wikipedia. Detailed explanation of ionic bond formation and properties. Wikipedia article
Covalent Bonding - Wikipedia. Comprehensive guide to electron sharing and molecular formation. Wikipedia article
Electronegativity - Wikipedia. Pauling scale and bond polarity predictions. Wikipedia article
Chemistry: The Central Science - Brown, LeMay, Bursten, Murphy, Woodward (2018). Standard chemistry textbook covering bonding theory. Pageplace.de
Materials Science and Engineering: An Introduction - Callister, W. D. & Rethwisch, D. G. (2018). Applications of ionic and covalent bonding in materials. Anupturnedworld
