

Executive Summary
Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol), calculated by summing the atomic masses of all atoms in a chemical formula. The key concept: molar mass provides the conversion factor between mass (grams) and amount of substance (moles), making it essential for all stoichiometric calculations in chemistry. One mole of any substance contains Avogadro's number (6.022 × 10²³) of particles, and the molar mass in grams numerically equals the atomic or molecular mass in atomic mass units (amu).
To calculate molar mass, identify all atoms in the chemical formula, find each element's atomic mass from the periodic table, multiply each atomic mass by the number of that atom present, and sum all values. For example, water (H₂O) has a molar mass of 18.02 g/mol calculated from 2 hydrogen atoms (2 × 1.008 g/mol) plus 1 oxygen atom (1 × 16.00 g/mol). This value means one mole of water molecules weighs 18.02 grams.
Understanding molar mass is fundamental for converting between mass and moles in chemical reactions, determining empirical and molecular formulas, calculating percent composition, and working with stoichiometry problems in materials science, pharmaceutical chemistry, and industrial processes.
Table of Contents
1. What is Molar Mass?
Molar mass is the mass of one mole of a substance, measured in grams per mole (g/mol). A mole is the SI unit for amount of substance, defined as exactly 6.022 × 10²³ particles (Avogadro's number). This number was chosen so that one mole of carbon-12 atoms has a mass of exactly 12 grams, creating a direct conversion between atomic mass units (used for individual atoms) and grams (used in the laboratory).
Units Explained: g/mol
The unit grams per mole (g/mol) indicates the mass in grams of one mole of particles. For any element, the molar mass in g/mol numerically equals the atomic mass in amu (atomic mass units) found on the periodic table. For example:
Carbon: atomic mass = 12.01 amu, molar mass = 12.01 g/mol
Oxygen: atomic mass = 16.00 amu, molar mass = 16.00 g/mol
Hydrogen: atomic mass = 1.008 amu, molar mass = 1.008 g/mol
This direct correspondence makes calculations straightforward. The periodic table atomic mass tells you both the mass of one atom in amu and the mass of one mole of atoms in grams.
Relationship to Atomic Mass
Atomic mass (found on the periodic table) represents the weighted average mass of all naturally occurring isotopes of an element, measured in atomic mass units. Molar mass uses these same atomic mass values but applies them to Avogadro's number of atoms, converting the unit from amu per atom to grams per mole.
Example: Chlorine has an atomic mass of 35.45 amu (weighted average of Cl-35 and Cl-37 isotopes). Therefore, one mole of chlorine atoms has a mass of 35.45 grams, giving chlorine a molar mass of 35.45 g/mol.
For molecules and compounds, molar mass is the sum of the molar masses of all constituent atoms. Water (H₂O) contains 2 hydrogen atoms and 1 oxygen atom, so its molar mass is the sum of the molar masses of these atoms.
2. How to Calculate Molar Mass
Step 1: Find Atomic Masses
Locate the atomic mass for each element in the chemical formula using the periodic table. Atomic masses appear below the element symbol, typically rounded to two or four decimal places depending on precision needed.
Common elements and their atomic masses:
H (Hydrogen): 1.008 amu
C (Carbon): 12.01 amu
N (Nitrogen): 14.01 amu
O (Oxygen): 16.00 amu
S (Sulfur): 32.07 amu
Cl (Chlorine): 35.45 amu
Na (Sodium): 22.99 amu
Ca (Calcium): 40.08 amu
For most calculations, rounding to two decimal places provides sufficient accuracy. Use more decimal places for precise analytical work or when small differences matter.
Step 2: Count Each Atom
Identify how many atoms of each element appear in the chemical formula. Subscripts (small numbers) following element symbols indicate the number of that atom present. When no subscript appears, assume one atom.
Reading chemical formulas:
H₂O: 2 hydrogen, 1 oxygen
CO₂: 1 carbon, 2 oxygen
Ca(NO₃)₂: 1 calcium, 2 nitrogen, 6 oxygen (parentheses mean the entire group inside repeats)
C₆H₁₂O₆: 6 carbon, 12 hydrogen, 6 oxygen
Parentheses rule: The subscript outside parentheses multiplies everything inside. In Ca(NO₃)₂, the subscript 2 means two complete NO₃ groups, giving 2 nitrogen atoms and 6 oxygen atoms total.
Step 3: Multiply and Add
For each element, multiply its atomic mass by the number of atoms present. Then sum all these values to get the total molar mass.
General formula: Molar Mass = Σ (number of atoms × atomic mass) for each element
Example - Carbon dioxide (CO₂):
Carbon: 1 atom × 12.01 g/mol = 12.01 g/mol
Oxygen: 2 atoms × 16.00 g/mol = 32.00 g/mol
Total molar mass: 12.01 + 32.00 = 44.01 g/mol
3. Worked Examples
Simple Molecules
Example 1: Water (H₂O)
Step 1: Find atomic masses
H = 1.008 g/mol
O = 16.00 g/mol
Step 2: Count atoms
2 hydrogen atoms
1 oxygen atom
Step 3: Calculate
Hydrogen: 2 × 1.008 = 2.016 g/mol
Oxygen: 1 × 16.00 = 16.00 g/mol
Total: 2.016 + 16.00 = 18.02 g/mol
Answer: The molar mass of water is 18.02 g/mol.
Example 2: Sodium chloride (NaCl)
Step 1: Find atomic masses
Na = 22.99 g/mol
Cl = 35.45 g/mol
Step 2: Count atoms
1 sodium atom
1 chlorine atom
Step 3: Calculate
Sodium: 1 × 22.99 = 22.99 g/mol
Chlorine: 1 × 35.45 = 35.45 g/mol
Total: 22.99 + 35.45 = 58.44 g/mol
Answer: The molar mass of sodium chloride is 58.44 g/mol.
Complex Molecules
Example 3: Glucose (C₆H₁₂O₆)
Step 1: Find atomic masses
C = 12.01 g/mol
H = 1.008 g/mol
O = 16.00 g/mol
Step 2: Count atoms
6 carbon atoms
12 hydrogen atoms
6 oxygen atoms
Step 3: Calculate
Carbon: 6 × 12.01 = 72.06 g/mol
Hydrogen: 12 × 1.008 = 12.10 g/mol
Oxygen: 6 × 16.00 = 96.00 g/mol
Total: 72.06 + 12.10 + 96.00 = 180.16 g/mol
Answer: The molar mass of glucose is 180.16 g/mol.
Example 4: Aspirin (C₉H₈O₄)
Step 1: Find atomic masses
C = 12.01 g/mol
H = 1.008 g/mol
O = 16.00 g/mol
Step 2: Count atoms
9 carbon atoms
8 hydrogen atoms
4 oxygen atoms
Step 3: Calculate
Carbon: 9 × 12.01 = 108.09 g/mol
Hydrogen: 8 × 1.008 = 8.064 g/mol
Oxygen: 4 × 16.00 = 64.00 g/mol
Total: 108.09 + 8.064 + 64.00 = 180.15 g/mol
Answer: The molar mass of aspirin is 180.15 g/mol.
Ionic Compounds with Polyatomic Ions
Example 5: Calcium nitrate (Ca(NO₃)₂)
Step 1: Find atomic masses
Ca = 40.08 g/mol
N = 14.01 g/mol
O = 16.00 g/mol
Step 2: Count atoms (careful with parentheses!)
1 calcium atom
2 nitrogen atoms (subscript 2 outside parentheses)
6 oxygen atoms (3 oxygen per NO₃ group × 2 groups)
Step 3: Calculate
Calcium: 1 × 40.08 = 40.08 g/mol
Nitrogen: 2 × 14.01 = 28.02 g/mol
Oxygen: 6 × 16.00 = 96.00 g/mol
Total: 40.08 + 28.02 + 96.00 = 164.10 g/mol
Answer: The molar mass of calcium nitrate is 164.10 g/mol.
4. Molar Mass vs Molecular Weight vs Formula Weight
Key Differences
These terms are often used interchangeably in general chemistry, but technically have distinct meanings:
Molar Mass:
Definition: Mass of one mole of a substance in grams per mole
Units: g/mol (always)
Applies to: Any substance (elements, molecules, ionic compounds)
Example: Molar mass of water = 18.02 g/mol
Molecular Weight:
Definition: Sum of atomic weights of atoms in a molecule
Units: amu (atomic mass units) or g/mol
Applies to: Only covalent molecules (not ionic compounds)
Example: Molecular weight of water = 18.02 amu
Formula Weight:
Definition: Sum of atomic weights of atoms in a formula unit
Units: amu or g/mol
Applies to: Ionic compounds (that don't form discrete molecules)
Example: Formula weight of NaCl = 58.44 amu
Important note: Numerically, these values are identical when expressed in their respective units. The molar mass in g/mol equals the molecular or formula weight in amu. The distinction is primarily about what type of substance you're describing and which units you're using.
When to Use Each Term
Use "molar mass" when:
Performing stoichiometric calculations (most common use)
Converting between mass and moles
Working with any type of substance
Units are g/mol
Use "molecular weight" when:
Discussing covalent molecules specifically
Describing relative molecular masses
Units could be amu or g/mol
Use "formula weight" when:
Discussing ionic compounds
Referring to the formula unit rather than molecules
Units could be amu or g/mol
In practice, "molar mass" is the most versatile term and appropriate for nearly all situations in general chemistry and stoichiometry.
6. Applications in Materials Science
Polymer Chemistry
Polymer molecular weight determines physical properties including strength, flexibility, melting point, and solubility. Chemists calculate number-average molecular weight (Mn) and weight-average molecular weight (Mw) to characterize polymer samples.
Polyethylene example: The repeat unit (CH₂CH₂) has molar mass 28.05 g/mol. A polyethylene chain with 1,000 repeat units has molecular weight approximately 28,050 g/mol. Higher molecular weight polymers generally have greater tensile strength and higher melting points.
Applications:
Plastics engineering (HDPE vs LDPE differences)
Biomedical polymers (degradation rates controlled by molecular weight)
Polymer processing (viscosity increases with molecular weight)
Drug Formulation
Pharmaceutical chemists use molar mass to calculate precise drug dosages, determine active ingredient concentrations, and design drug delivery systems.
Aspirin dosage calculation: Aspirin (C₉H₈O₄) has molar mass 180.15 g/mol. A standard 325 mg tablet contains: 325 mg ÷ 180.15 g/mol = 0.001804 mol = 1.804 mmol of aspirin
This corresponds to 1.09 × 10²¹ molecules of aspirin (using Avogadro's number), demonstrating how molar mass connects macroscopic dosages to molecular quantities.
Applications:
Formulation development (calculating excipient ratios)
Quality control (verifying active ingredient content)
Pharmacokinetics (modeling drug metabolism rates)
Battery Materials
Lithium-ion battery performance depends on precise stoichiometry of electrode materials. Molar mass calculations ensure correct lithium-to-metal ratios in cathode materials.
LiCoO₂ cathode example:
Li = 6.94 g/mol
Co = 58.93 g/mol
O = 16.00 g/mol
Total molar mass = 6.94 + 58.93 + (2 × 16.00) = 97.87 g/mol
Theoretical capacity calculations use molar mass to determine maximum lithium storage: Theoretical capacity = (F × number of electrons) ÷ (3.6 × molar mass) where F = Faraday's constant
Applications:
Electrode material synthesis (precise Li:Co:O ratios)
Capacity calculations (predicting battery performance)
Electrolyte formulation (molarity calculations)
7. Common Mistakes to Avoid
Mistake 1: Forgetting to Multiply by Subscripts
Problem: Counting atoms incorrectly, especially with subscripts or parentheses.
Wrong: Calculating H₂O as 1.008 + 16.00 = 17.01 g/mol (forgot to multiply H by 2)
Correct: H₂O = (2 × 1.008) + 16.00 = 18.02 g/mol
Solution: Write out the multiplication explicitly for each element before adding. For H₂O, write "2 × 1.008 = 2.016" as a separate step.
Mistake 2: Mishandling Parentheses in Chemical Formulas
Problem: Not multiplying the subscript outside parentheses by all atoms inside.
Example: Ca(NO₃)₂
Wrong: Counting as 1 Ca, 1 N, 3 O (only used the subscript inside parentheses)
Correct: 1 Ca, 2 N, 6 O (subscript 2 outside multiplies both N and O₃)
Solution: Expand formulas with parentheses before calculating. Write Ca(NO₃)₂ as CaN₂O₆ mentally to avoid errors.
Mistake 3: Using Wrong Atomic Masses
Problem: Reading atomic number instead of atomic mass, or using outdated values.
Wrong: Using 6 for carbon (atomic number) instead of 12.01 (atomic mass)
Correct: Always use atomic mass from periodic table, typically the larger number below the element symbol.
Solution: Double-check which number you're reading. Atomic number (top) is always a whole number. Atomic mass (bottom) has decimal places and represents weighted average of isotopes.
Mistake 4: Rounding Too Early
Problem: Rounding intermediate values causes final answer to be inaccurate.
Wrong calculation for C₆H₁₂O₆:
C: 6 × 12 = 72 (rounded 12.01 to 12)
H: 12 × 1 = 12 (rounded 1.008 to 1)
O: 6 × 16 = 96 (exact)
Total = 180 g/mol (actual: 180.16 g/mol)
Correct: Keep full precision until the final answer, then round.
Solution: Use full atomic mass values throughout calculation. Only round the final molar mass to appropriate significant figures (usually 2-4 decimal places).
8. FAQ
What is molar mass?
Molar mass is the mass of one mole of a substance, measured in grams per mole (g/mol). One mole contains Avogadro's number (6.022 × 10²³) of particles, whether atoms, molecules, or formula units. Molar mass provides the conversion factor between mass (grams) and amount of substance (moles), making it essential for stoichiometric calculations. For any element, the molar mass in g/mol numerically equals the atomic mass in atomic mass units (amu) found on the periodic table. For compounds, molar mass is calculated by summing the atomic masses of all constituent atoms. Water (H₂O) has a molar mass of 18.02 g/mol, meaning one mole of water molecules weighs 18.02 grams. This fundamental concept connects the microscopic world of atoms and molecules to macroscopic quantities we can measure in the laboratory.
How do you calculate molar mass?
Calculate molar mass in three steps: (1) Find the atomic mass for each element from the periodic table. (2) Count how many atoms of each element appear in the chemical formula, paying careful attention to subscripts and parentheses. (3) Multiply each element's atomic mass by its number of atoms, then sum all values. For example, to find the molar mass of carbon dioxide (CO₂): carbon has atomic mass 12.01 g/mol and appears once (1 × 12.01 = 12.01), oxygen has atomic mass 16.00 g/mol and appears twice (2 × 16.00 = 32.00), total molar mass = 12.01 + 32.00 = 44.01 g/mol. For compounds with parentheses like Ca(NO₃)₂, the subscript outside multiplies everything inside, giving 1 Ca, 2 N, and 6 O atoms. Write out each calculation explicitly to avoid errors.
What is the difference between molar mass and molecular weight?
Molar mass and molecular weight are numerically identical but differ in units and technical meaning. Molar mass is the mass of one mole of a substance expressed in grams per mole (g/mol), while molecular weight is the sum of atomic weights in a molecule expressed in atomic mass units (amu). Molar mass applies to any substance and is the preferred term for stoichiometric calculations involving mass-to-mole conversions. Molecular weight technically applies only to covalent molecules, not ionic compounds. Formula weight is used for ionic compounds that don't form discrete molecules. For water, molar mass = 18.02 g/mol and molecular weight = 18.02 amu - same number, different units. In practice, "molar mass" is the most versatile term and appropriate for nearly all chemistry calculations since it directly relates laboratory measurements (grams) to chemical amounts (moles).
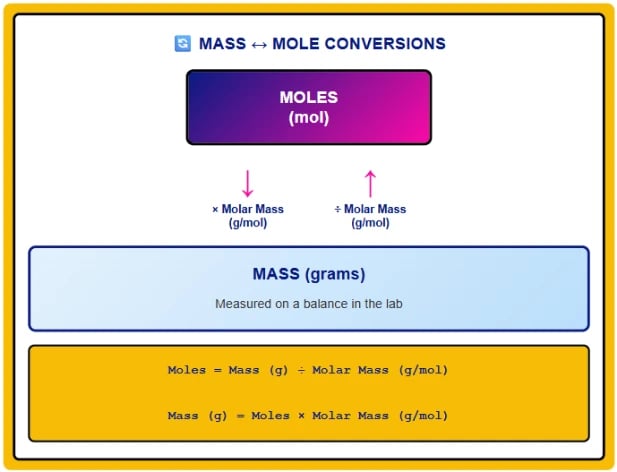
How do you convert mass to moles using molar mass?
Convert mass to moles by dividing the mass in grams by the molar mass in g/mol. The formula is: Moles = Mass (g) ÷ Molar Mass (g/mol). For example, to find how many moles are in 50.0 g of sodium chloride (NaCl): First, calculate molar mass of NaCl = 22.99 + 35.45 = 58.44 g/mol. Then apply the formula: Moles = 50.0 g ÷ 58.44 g/mol = 0.856 mol. Notice the gram units cancel, leaving only moles. This conversion is fundamental to stoichiometry because chemical equations give mole ratios, not mass ratios. To use balanced equations for predictions, you must first convert given masses to moles, apply mole ratios from the equation, then convert back to masses using molar mass. The molar mass serves as the bridge between measurable quantities (grams) and chemical quantities (moles).
Why does molar mass equal atomic mass numerically?
Molar mass in g/mol equals atomic mass in amu numerically because of how the mole and atomic mass unit are defined. One atomic mass unit (amu) is defined as exactly 1/12 the mass of a carbon-12 atom. Avogadro's number (6.022 × 10²³) was chosen so that one mole of carbon-12 atoms has a mass of exactly 12 grams. This creates a direct correspondence: an element with atomic mass X amu has a molar mass of X g/mol. For example, oxygen has atomic mass 16.00 amu because each oxygen atom weighs 16.00 times more than 1/12 of a carbon-12 atom. One mole of oxygen atoms (6.022 × 10²³ atoms) weighs exactly 16.00 grams, giving molar mass of 16.00 g/mol. This elegant relationship allows chemists to use periodic table values for both individual atom masses (in amu) and mole quantities (in g/mol) interchangeably.
What units are used for molar mass?
Molar mass is always expressed in grams per mole (g/mol). This unit indicates the mass in grams of one mole (6.022 × 10²³ particles) of a substance. The g/mol unit is fundamental to stoichiometric calculations because it provides the conversion factor between mass (which we measure on a balance) and moles (which appear in chemical equations). Some older texts use kg/kmol, which is numerically identical to g/mol. Molecular weight uses atomic mass units (amu) instead of g/mol, but the numerical values are the same. For example, water has molar mass 18.02 g/mol, meaning 6.022 × 10²³ water molecules collectively weigh 18.02 grams. Never use units like g/molecule or kg/mol for molar mass in standard chemistry calculations. Always use g/mol for molar mass, and always include the unit in your answer to avoid confusion with other mass-related quantities.
Related Articles
Continue exploring chemical calculations and materials science:
Stoichiometry: Complete Guide with Examples - Using molar mass in calculations
Polyatomic Ions: Complete List & Guide - Calculating formula weights
Ionic vs Covalent Bonds: Complete Guide - Understanding molecular compounds
Sources and References
Molar Mass - Wikipedia. Definition, calculation methods, and applications. Wikipedia article
Atomic Mass - Wikipedia. Relationship between atomic mass and molar mass. Wikipedia article
Mole (unit) - Wikipedia. Definition of the mole and Avogadro's number. Wikipedia article
Chemistry: The Science in Context - Thomas R. Gilbert, Rein V. Kirss, Natalie Foster, Stacey Lowery Bretz. General chemistry textbook covering molar mass calculations and stoichiometry. Free PDF
The Role of Industrial Chemistry in Modern Manufacturing - Applications of molar mass in chemical manufacturing and materials science. Open Access PDF
