Executive Summary
Polyatomic ions are groups of two or more covalently bonded atoms that carry a net electric charge and act as a single unit in chemical reactions and ionic compounds. The key concept: unlike monatomic ions (single atoms with charge), polyatomic ions contain multiple atoms bonded together that collectively possess positive or negative charge and remain intact during most chemical reactions. Common examples include sulfate (SO₄²⁻), nitrate (NO₃⁻), ammonium (NH₄⁺), and carbonate (CO₃²⁻).
Understanding polyatomic ions is essential for writing chemical formulas, naming ionic compounds, predicting reaction products, and working with materials science applications. Most polyatomic ions are anions (negatively charged), with only a few common cations (positively charged). When writing formulas with multiple polyatomic ions, parentheses are required to show that the entire group repeats as a unit.
This guide provides a complete list of common polyatomic ions organized by charge, memorization strategies proven to work for students, naming conventions following IUPAC rules, and step-by-step methods for writing chemical formulas correctly.
Table of Contents
1. What are Polyatomic Ions?
A polyatomic ion is a charged molecular ion composed of two or more atoms covalently bonded together, functioning as a single charged unit in chemical reactions and compounds. The prefix "poly" means "many," indicating multiple atoms, while "atomic" refers to the constituent atoms within the ion. Unlike monatomic ions formed from single atoms gaining or losing electrons (Na⁺, Cl⁻), polyatomic ions maintain their multi-atom structure across most chemical processes.
Structure and Bonding
Within a polyatomic ion, atoms connect through covalent bonds where electrons are shared between atoms. However, the entire group carries a net electric charge because the total number of electrons does not equal the total number of protons across all atoms in the ion. This combination of covalent bonding within the ion and ionic behavior in compounds makes polyatomic ions unique.
Example - Sulfate Ion (SO₄²⁻):
One sulfur atom bonded to four oxygen atoms
Covalent S-O bonds within the ion
Overall charge of 2- (two more electrons than protons)
Acts as a single unit with 2- charge
Difference from Monatomic Ions
Monatomic ions consist of single atoms with charge:
Na⁺ (sodium loses 1 electron)
Cl⁻ (chlorine gains 1 electron)
Ca²⁺ (calcium loses 2 electrons)
Polyatomic ions consist of bonded atom groups with charge:
SO₄²⁻ (sulfate: 1 S + 4 O atoms, 2- charge)
NO₃⁻ (nitrate: 1 N + 3 O atoms, 1- charge)
NH₄⁺ (ammonium: 1 N + 4 H atoms, 1+ charge)
Why Polyatomic Ions Stay Together
The covalent bonds holding atoms together within polyatomic ions are strong, requiring significant energy to break. During typical chemical reactions, ionic compounds dissolve or react, but polyatomic ions usually remain intact as units. For example, when calcium nitrate Ca(NO₃)₂ dissolves in water, it separates into Ca²⁺ and two NO₃⁻ ions. The nitrate ions do not break apart into nitrogen and oxygen atoms under normal conditions.
Only under extreme conditions (very high temperatures, strong acids or bases, electrochemical reactions) do polyatomic ions decompose into their constituent atoms. In ordinary chemistry lab reactions, you can safely treat polyatomic ions as indivisible charged units.
2. Complete List of Common Polyatomic Ions
Negative Polyatomic Ions (Anions)
Most polyatomic ions are anions, carrying negative charges. These form when atom groups gain extra electrons or when molecules lose protons (H⁺ ions), leaving behind negative charge.
Essential Polyatomic Anions (Must Memorize):
1- Charge:
Acetate: C₂H₃O₂⁻ or CH₃COO⁻
Nitrate: NO₃⁻
Nitrite: NO₂⁻
Hydroxide: OH⁻
Cyanide: CN⁻
Permanganate: MnO₄⁻
Hydrogen carbonate (bicarbonate): HCO₃⁻
Hydrogen sulfate (bisulfate): HSO₄⁻
Dihydrogen phosphate: H₂PO₄⁻
Chlorate: ClO₃⁻
Chlorite: ClO₂⁻
Hypochlorite: ClO⁻
Perchlorate: ClO₄⁻
2- Charge:
Sulfate: SO₄²⁻
Sulfite: SO₃²⁻
Carbonate: CO₃²⁻
Chromate: CrO₄²⁻
Dichromate: Cr₂O₇²⁻
Oxalate: C₂O₄²⁻
Hydrogen phosphate: HPO₄²⁻
Peroxide: O₂²⁻
3- Charge:
Phosphate: PO₄³⁻
Phosphite: PO₃³⁻
Positive Polyatomic Ions (Cations)
Only a few common polyatomic ions carry positive charges. These form when atom groups lose electrons or when molecules gain protons (H⁺ ions).
Common Polyatomic Cations:
1+ Charge:
Ammonium: NH₄⁺
Hydronium: H₃O⁺
2+ Charge:
Mercury(I): Hg₂²⁺ (special case: two mercury atoms bonded, total 2+ charge)
The ammonium ion (NH₄⁺) is by far the most important polyatomic cation to memorize. It appears in countless compounds including ammonium nitrate (fertilizer/explosive), ammonium chloride (batteries), and ammonium sulfate (fertilizer). The hydronium ion (H₃O⁺) is crucial in acid-base chemistry, representing the form of H⁺ ions in aqueous solutions.
3. Memorization Strategies
Pattern Recognition Method
Oxygen Series (ate/ite patterns):
Many polyatomic ions contain oxygen. For ions with the same central atom but different numbers of oxygen atoms, systematic naming helps memorization:
Most oxygen → Least oxygen:
per____ate (most oxygen): ClO₄⁻ (perchlorate)
____ate (more oxygen): ClO₃⁻ (chlorate)
____ite (fewer oxygen): ClO₂⁻ (chlorite)
hypo____ite (least oxygen): ClO⁻ (hypochlorite)
This pattern applies to chlorine, bromine, and iodine oxyanions. Memorize one series (chlorine), and the pattern transfers to others.
Sulfur and Nitrogen Series:
SO₄²⁻ (sulfate) has more O than SO₃²⁻ (sulfite)
NO₃⁻ (nitrate) has more O than NO₂⁻ (nitrite)
Rule: -ate ending indicates more oxygen atoms than -ite ending for the same element.
Mnemonic Devices
"Nick the Camel ate a Clam for Supper in Phoenix"
This classic mnemonic encodes charges and number of atoms:
Nick = Nitrate, NO₃⁻ (1- charge, 3 oxygens)
Camel = Carbonate, CO₃²⁻ (2- charge, 3 oxygens)
Clam = Chlorate, ClO₃⁻ (1- charge, 3 oxygens)
Supper = Sulfate, SO₄²⁻ (2- charge, 4 oxygens)
Phoenix = Phosphate, PO₄³⁻ (3- charge, 4 oxygens)
Consonants (N, C, C, S, P) give the elements. Vowels encode charges and oxygen counts.
Common Ion Rhyme: "Hydroxide is OH with minus one, Nitrate is NO₃ when all is done, Sulfate takes SO₄ with charge of two, Phosphate needs PO₄ with minus three for you."
Grouping by Element
Organize ions by central atom:
Nitrogen ions:
NO₃⁻ (nitrate)
NO₂⁻ (nitrite)
Sulfur ions:
SO₄²⁻ (sulfate)
SO₃²⁻ (sulfite)
HSO₄⁻ (hydrogen sulfate)
Phosphorus ions:
PO₄³⁻ (phosphate)
HPO₄²⁻ (hydrogen phosphate)
H₂PO₄⁻ (dihydrogen phosphate)
Carbon ions:
CO₃²⁻ (carbonate)
HCO₃⁻ (hydrogen carbonate/bicarbonate)
C₂H₃O₂⁻ (acetate)
Chlorine ions:
ClO₄⁻ (perchlorate)
ClO₃⁻ (chlorate)
ClO₂⁻ (chlorite)
ClO⁻ (hypochlorite)
Grouping by element reveals patterns in charge and structure, making memorization more systematic.
4. Naming Polyatomic Ions
-ate vs -ite Endings
The -ate and -ite suffixes distinguish between polyatomic ions containing oxygen (oxyanions) with the same central atom but different oxygen counts.
General Rule:
-ate ending: More oxygen atoms (sulfate SO₄²⁻ has 4 oxygens)
-ite ending: Fewer oxygen atoms (sulfite SO₃²⁻ has 3 oxygens)
Common Pairs:
-ate (more O)-ite (fewer O)Sulfate SO₄²⁻Sulfite SO₃²⁻Nitrate NO₃⁻Nitrite NO₂⁻Phosphate PO₄³⁻Phosphite PO₃³⁻Chlorate ClO₃⁻Chlorite ClO₂⁻
The actual number of oxygen atoms is not absolute across different elements. Sulfate has 4 oxygens while nitrate has 3, but both use -ate because they represent the "more oxygen" form for their respective elements.
Per- and Hypo- Prefixes
When an element forms more than two oxyanions, prefixes expand the naming system beyond -ate/-ite:
Four-ion series (halogens: Cl, Br, I):
per-____-ate: Most oxygen (perchlorate ClO₄⁻)
____-ate: More oxygen (chlorate ClO₃⁻)
____-ite: Fewer oxygen (chlorite ClO₂⁻)
hypo-____-ite: Least oxygen (hypochlorite ClO⁻)
Examples:
Perchlorate ClO₄⁻: "per" = above/beyond, most oxygen
Hypochlorite ClO⁻: "hypo" = below/under, least oxygen
Permanganate (MnO₄⁻) is another example using "per-" prefix for high oxygen content.
Hydrogen Additions
Adding hydrogen atoms to polyatomic anions reduces negative charge by one per hydrogen added and changes the name by adding "hydrogen" or "dihydrogen" prefix.
Carbonate Series:
CO₃²⁻: Carbonate (2- charge, no H)
HCO₃⁻: Hydrogen carbonate or bicarbonate (1- charge, one H added)
Phosphate Series:
PO₄³⁻: Phosphate (3- charge, no H)
HPO₄²⁻: Hydrogen phosphate (2- charge, one H added)
H₂PO₄⁻: Dihydrogen phosphate (1- charge, two H added)
Sulfate Series:
SO₄²⁻: Sulfate (2- charge, no H)
HSO₄⁻: Hydrogen sulfate or bisulfate (1- charge, one H added)
The "bi-" prefix is an older naming convention (bicarbonate, bisulfate) equivalent to "hydrogen" prefix. Modern IUPAC nomenclature prefers "hydrogen carbonate" over "bicarbonate," but both names remain widely used.
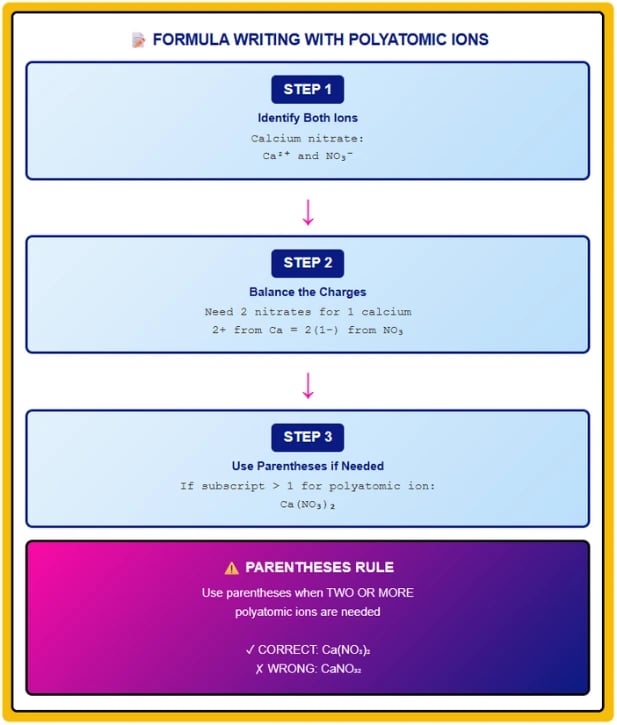
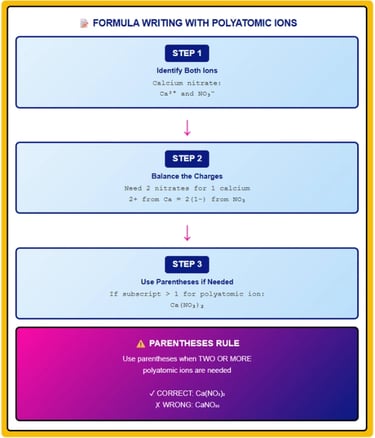
5. Writing Chemical Formulas with Polyatomic Ions
Single Polyatomic Ion
When one polyatomic ion combines with a monatomic ion, write the formula directly without parentheses.
Steps:
Write cation symbol first (positive ion)
Write anion symbol second (negative ion)
Balance charges using subscripts
Example 1: Sodium nitrate
Sodium: Na⁺ (1+ charge)
Nitrate: NO₃⁻ (1- charge)
Charges balance 1:1
Formula: NaNO₃
Example 2: Calcium sulfate
Calcium: Ca²⁺ (2+ charge)
Sulfate: SO₄²⁻ (2- charge)
Charges balance 1:1
Formula: CaSO₄
Example 3: Aluminum nitrate
Aluminum: Al³⁺ (3+ charge)
Nitrate: NO₃⁻ (1- charge)
Need three nitrates to balance one aluminum
Formula: Al(NO₃)₃ (parentheses needed - see next section)
Multiple Polyatomic Ions (Parentheses Rule)
CRITICAL RULE: When two or more polyatomic ions are needed in a formula, enclose the polyatomic ion in parentheses and place the subscript outside.
Why parentheses matter:
Without: Al(NO₃)₃ correctly shows three nitrate ions
Wrong: AlNO₃₃ incorrectly suggests thirty-three oxygen atoms
Parentheses indicate the entire group repeats, not just the last element.
Example 1: Calcium nitrate
Calcium: Ca²⁺ (2+ charge)
Nitrate: NO₃⁻ (1- charge)
Need two nitrates to balance calcium
Formula: Ca(NO₃)₂
The subscript 2 applies to the entire NO₃ group, giving 2 nitrogen atoms and 6 oxygen atoms total.
Example 2: Ammonium sulfate
Ammonium: NH₄⁺ (1+ charge)
Sulfate: SO₄²⁻ (2- charge)
Need two ammoniums to balance sulfate
Formula: (NH₄)₂SO₄
Example 3: Iron(III) phosphate
Iron(III): Fe³⁺ (3+ charge)
Phosphate: PO₄³⁻ (3- charge)
Charges balance 1:1
Formula: FePO₄ (no parentheses needed - only one phosphate)
Worked Examples
Problem 1: Write the formula for magnesium hydroxide
Step 1: Identify ions
Magnesium: Mg²⁺
Hydroxide: OH⁻
Step 2: Balance charges
Need two OH⁻ to balance Mg²⁺
Total positive: 2+
Total negative: 2(1-) = 2-
Step 3: Write formula with parentheses
Mg(OH)₂
Problem 2: Write the formula for ammonium carbonate
Step 1: Identify ions
Ammonium: NH₄⁺
Carbonate: CO₃²⁻
Step 2: Balance charges
Need two NH₄⁺ to balance CO₃²⁻
Total positive: 2(1+) = 2+
Total negative: 2-
Step 3: Write formula with parentheses
(NH₄)₂CO₃
Problem 3: Write the formula for calcium phosphate
Step 1: Identify ions
Calcium: Ca²⁺
Phosphate: PO₄³⁻
Step 2: Balance charges (least common multiple method)
LCM of 2 and 3 is 6
Need three Ca²⁺ (3 × 2+ = 6+)
Need two PO₄³⁻ (2 × 3- = 6-)
Step 3: Write formula with parentheses
Ca₃(PO₄)₂
6. Common Polyatomic Compounds
In Everyday Life
Sodium bicarbonate (NaHCO₃) - Baking soda: Contains hydrogen carbonate (HCO₃⁻) polyatomic ion. Used in cooking, cleaning, antacids, and fire extinguishers. When heated or mixed with acid, decomposes to release CO₂ gas, causing baked goods to rise.
Calcium carbonate (CaCO₃) - Limestone, chalk, antacids: Contains carbonate (CO₃²⁻) ion. Major component of sedimentary rocks, seashells, and coral reefs. Used in construction (cement, concrete), agriculture (soil pH adjustment), and medicine (calcium supplements).
Ammonium nitrate (NH₄NO₃) - Fertilizer: Contains both ammonium (NH₄⁺) cation and nitrate (NO₃⁻) anion. Provides nitrogen for plant growth. Also used as explosive component due to rapid decomposition releasing gases and energy.
Sodium hypochlorite (NaClO) - Bleach: Contains hypochlorite (ClO⁻) ion. Powerful oxidizing agent used for disinfection, water treatment, and stain removal. Active ingredient in household bleach solutions.
In Materials Science
Lithium iron phosphate (LiFePO₄) - Battery cathodes: Contains phosphate (PO₄³⁻) ion. Used in lithium-ion batteries for electric vehicles and energy storage systems. Offers thermal stability, long cycle life, and safety advantages over cobalt-based cathodes.
Calcium phosphate (Ca₃(PO₄)₂) - Bioceramics: Contains phosphate ion. Major mineral component of bones and teeth (hydroxyapatite Ca₁₀(PO₄)₆(OH)₂). Used in bone grafts, dental implants, and tissue engineering scaffolds due to biocompatibility and osteoconductive properties.
Barium sulfate (BaSO₄) - X-ray contrast agent: Contains sulfate (SO₄²⁻) ion. Insoluble in water, making it safe for medical imaging. Patients ingest barium sulfate suspension before X-rays or CT scans to visualize digestive system. Also used as white pigment in paints and as weighting agent in oil drilling fluids.
Ammonium perchlorate (NH₄ClO₄) - Rocket propellant: Contains ammonium and perchlorate (ClO₄⁻) ions. Powerful oxidizer in solid rocket boosters, including Space Shuttle boosters and military missiles. Decomposes at high temperatures, releasing oxygen to combust fuel.
In Pharmaceuticals
Magnesium sulfate (MgSO₄) - Epsom salt: Contains sulfate ion. Used medically to treat magnesium deficiency, eclampsia in pregnancy, and as laxative. Dissolved in bathwater for muscle relaxation and pain relief.
Sodium nitrite (NaNO₂) - Food preservative: Contains nitrite (NO₂⁻) ion. Prevents bacterial growth in cured meats (bacon, ham, hot dogs). Also used as antidote for cyanide poisoning, forming methemoglobin that binds cyanide.
Potassium permanganate (KMnO₄) - Antiseptic: Contains permanganate (MnO₄⁻) ion. Strong oxidizing agent with antimicrobial properties. Used topically to treat fungal infections, wounds, and dermatitis. Also employed in water treatment to remove iron and manganese.
7. Common Mistakes to Avoid
Mistake 1: Breaking Apart Polyatomic Ions
Problem: Treating polyatomic ions as separate atoms instead of a single unit.
Wrong approach: "Calcium nitrate contains Ca, N, and O atoms, so I'll write CaNO₃."
Why it's wrong: This suggests one nitrogen and three oxygen atoms, but you need TWO nitrate ions to balance calcium's 2+ charge.
Correct approach: Recognize NO₃⁻ as indivisible unit. Two nitrates needed: Ca(NO₃)₂.
Solution: Memorize polyatomic ions as complete units. When balancing formulas, multiply the entire ion, not individual atoms within it.
Mistake 2: Forgetting Parentheses with Multiple Polyatomic Ions
Problem: Omitting parentheses when multiple polyatomic ions are needed.
Wrong: Mg(OH)₂ written as MgOH₂
Why it's wrong: MgOH₂ suggests one oxygen and two hydrogens (water), not two hydroxide ions.
Correct: Mg(OH)₂ clearly shows two complete OH⁻ units.
Solution: Use parentheses whenever the subscript for a polyatomic ion is greater than 1. Exception: when subscript is 1, parentheses are optional (CaSO₄ or Ca(SO₄) both correct, but CaSO₄ preferred).
Mistake 3: Confusing -ate and -ite Ion Names
Problem: Mixing up which ion has more oxygen.
Example: Writing sodium sulfite formula as Na₂SO₄ instead of Na₂SO₃.
Why it happens: Students remember there's a difference but forget which is which.
Solution: Remember "-ate has MORE oxygen" (alphabetically, "a" comes after "i," and -ate has more oxygen than -ite). Or use mnemonic: "I ate more, so -ate has more oxygen."
Verification: Sulfate SO₄²⁻ (4 oxygens) vs Sulfite SO₃²⁻ (3 oxygens). Nitrate NO₃⁻ (3 oxygens) vs Nitrite NO₂⁻ (2 oxygens).
Mistake 4: Incorrectly Adding Hydrogen to Polyatomic Ions
Problem: Not understanding how hydrogen addition affects charge.
Wrong: Thinking H₂CO₃ is hydrogen carbonate (it's actually carbonic acid, not an ion).
Correct series:
CO₃²⁻: Carbonate (2- charge)
HCO₃⁻: Hydrogen carbonate (1- charge, lost one negative)
H₂CO₃: Carbonic acid (neutral molecule, lost both negatives)
Solution: Each added hydrogen reduces negative charge by one. If all negative charges are neutralized, you have a neutral molecule (acid), not an ion.
8. FAQ
What are polyatomic ions?
Polyatomic ions are groups of two or more covalently bonded atoms that carry a net electric charge and function as a single unit in chemical reactions and ionic compounds. Unlike monatomic ions formed from single atoms, polyatomic ions maintain their multi-atom structure during most chemical processes. Common examples include sulfate (SO₄²⁻), nitrate (NO₃⁻), ammonium (NH₄⁺), and carbonate (CO₃²⁻). Within the ion, atoms connect through covalent bonds, but the entire group possesses overall positive or negative charge. Most polyatomic ions are anions (negatively charged), with only a few common cations like ammonium. These ions are essential in chemistry because they appear in countless compounds used in everyday life, industry, materials science, and pharmaceuticals.
How many polyatomic ions should I memorize?
For general chemistry courses, students should memorize approximately 15-20 polyatomic ions that appear frequently in problems and real-world applications. Essential ions include: sulfate (SO₄²⁻), sulfite (SO₃²⁻), nitrate (NO₃⁻), nitrite (NO₂⁻), carbonate (CO₃²⁻), hydrogen carbonate/bicarbonate (HCO₃⁻), phosphate (PO₄³⁻), hydroxide (OH⁻), ammonium (NH₄⁺), acetate (C₂H₃O₂⁻), chlorate (ClO₃⁻), perchlorate (ClO₄⁻), permanganate (MnO₄⁻), chromate (CrO₄²⁻), and dichromate (Cr₂O₇²⁻). Advanced courses may require additional ions. Focus first on the most common ions with 1- and 2- charges, then expand to 3- charges and less common ions. Using pattern recognition (ate/ite endings, per/hypo prefixes) and mnemonic devices significantly reduces pure memorization needed.
What is the difference between -ate and -ite polyatomic ions?
The -ate and -ite suffixes distinguish polyatomic ions containing oxygen (oxyanions) with the same central atom but different numbers of oxygen atoms. The -ate ending indicates more oxygen atoms, while the -ite ending indicates fewer oxygen atoms. For example, sulfate (SO₄²⁻) contains four oxygen atoms while sulfite (SO₃²⁻) contains three oxygen atoms. Similarly, nitrate (NO₃⁻) has more oxygen than nitrite (NO₂⁻). This pattern applies consistently across different elements, though the absolute number of oxygen atoms varies by element. Both ions typically have the same charge despite different oxygen counts. Remembering "-ate has MORE" helps avoid confusion. When elements form more than two oxyanions, prefixes expand the system: per- (most oxygen), -ate (more), -ite (fewer), hypo- (least oxygen), as seen in the chlorine series: perchlorate (ClO₄⁻), chlorate (ClO₃⁻), chlorite (ClO₂⁻), hypochlorite (ClO⁻).
When do I use parentheses in chemical formulas?
Use parentheses in chemical formulas when two or more polyatomic ions are needed to balance charges. Parentheses indicate that the entire polyatomic ion group repeats, not just the last element. For example, calcium nitrate requires two nitrate ions (NO₃⁻) to balance calcium's 2+ charge, written as Ca(NO₃)₂. The subscript 2 outside parentheses means two complete NO₃ units (2 nitrogen atoms and 6 oxygen atoms total). Without parentheses, CaNO₃₂ would incorrectly suggest 32 oxygen atoms. Parentheses are NOT needed when only one polyatomic ion is present (CaSO₄, not Ca(SO₄)) or when the subscript would be 1. Exception: When both ions are polyatomic and multiple copies needed, use parentheses for both, such as (NH₄)₂CO₃ for ammonium carbonate (two ammonium ions, one carbonate ion).
What is the most common polyatomic cation?
Ammonium (NH₄⁺) is by far the most common and important polyatomic cation. It consists of one nitrogen atom bonded to four hydrogen atoms with an overall 1+ charge. Ammonium forms when ammonia (NH₃) gains a proton (H⁺). This ion appears in numerous compounds including ammonium nitrate (NH₄NO₃) used as fertilizer and explosive, ammonium chloride (NH₄Cl) in batteries and cleaning products, and ammonium sulfate ((NH₄)₂SO₄) as fertilizer. Hydronium (H₃O⁺) is the only other commonly encountered polyatomic cation, representing the form of H⁺ ions in aqueous solution and playing a crucial role in acid-base chemistry. Mercury(I) (Hg₂²⁺) is a special case consisting of two bonded mercury atoms with total 2+ charge, but appears less frequently than ammonium in general chemistry.
How do I remember polyatomic ion charges?
Several strategies help remember polyatomic ion charges. First, use pattern recognition: most polyatomic ions are anions (negative), with ammonium being the main cation exception. Second, learn groups by element - all chlorine oxyanions have 1- charge (perchlorate, chlorate, chlorite, hypochlorite), sulfur oxyanions have 2- charge (sulfate, sulfite), and phosphorus oxyanions have 3- charge (phosphate). Third, use mnemonics like "Nick the Camel" which encodes both formulas and charges. Fourth, notice that adding hydrogen reduces negative charge by one: carbonate (CO₃²⁻) becomes hydrogen carbonate (HCO₃⁻) with 1- charge. Fifth, practice writing common compound formulas until charges become automatic - if you know calcium sulfate is CaSO₄, you remember sulfate must be 2- to balance calcium's 2+. Flashcards, repeated practice problems, and pattern recognition work better than pure memorization of isolated charges.
Related Articles
Continue exploring chemical bonding and materials science:
Ionic vs Covalent Bonds: Complete Guide - Understanding bonds in ionic compounds
Stoichiometry: Complete Guide with Examples - Calculate formulas with polyatomic ions
Stratified vs Cluster Sampling: Data Science Guide - Statistical methods for materials research
Sources and References
Polyatomic Ion - Wikipedia. Definition, common examples, and nomenclature. Wikipedia article
Chemical Nomenclature - Wikipedia. IUPAC naming conventions for ions and compounds. Wikipedia article
Oxyanion - Wikipedia. Oxygen-containing polyatomic anions and naming patterns. Wikipedia article
Chemistry: The Science in Context - Thomas R. Gilbert, Rein V. Kirss, Natalie Foster, Stacey Lowery Bretz. General chemistry textbook covering stoichiometry and mole concepts. Ung.edu
The Role of Industrial Chemistry in Modern Manufacturing - Applications of stoichiometry in chemical manufacturing and industrial processes. Omicsonline.org
