

Executive Summary
Stoichiometry is the quantitative study of reactants and products in chemical reactions, calculating the exact amounts of substances consumed and produced based on balanced chemical equations. The key concept: mole ratios from balanced equations allow you to convert between masses, moles, and volumes of different substances in a reaction. One mole of any substance contains Avogadro's number (6.022 × 10²³) of particles and has a mass equal to its molar mass in grams.
Use stoichiometry to predict how much product forms from given reactants, identify limiting reactants that determine maximum product yield, and calculate theoretical versus actual yields in real-world processes. The fundamental method involves four steps: write a balanced equation, convert given quantities to moles, apply mole ratios, and convert back to desired units. Understanding stoichiometry is essential for chemical manufacturing, pharmaceutical synthesis, materials engineering, and environmental chemistry.
This guide provides step-by-step calculation methods with worked examples, helping you master stoichiometric conversions and apply them to real-world problems in advanced materials and chemical processes.
Table of Contents
1. What is Stoichiometry?
Stoichiometry derives from Greek words meaning "element" and "measure," referring to the mathematical relationships between quantities of reactants and products in chemical reactions. A balanced chemical equation provides the recipe for a reaction, showing the exact proportions in which substances combine and form products.
Example: The combustion of methane: CH₄ + 2O₂ → CO₂ + 2H₂O
This equation tells us that 1 mole of methane reacts with 2 moles of oxygen to produce 1 mole of carbon dioxide and 2 moles of water. Stoichiometry allows us to scale these mole ratios to any quantity - grams, kilograms, tons - making it indispensable for industrial chemistry, pharmaceutical manufacturing, and materials synthesis.
Why Stoichiometry Matters
Every chemical process requires precise control of reactant amounts. Too much of one reactant wastes materials and money. Too little means incomplete reactions and reduced product yield. Stoichiometry provides the mathematical framework to optimize chemical processes, ensure consistent product quality, and minimize waste in industries ranging from drug synthesis to battery manufacturing.
In materials science, stoichiometry determines the exact composition of alloys, ceramics, and semiconductors. A silicon wafer for computer chips requires precise stoichiometric ratios of dopants (phosphorus, boron) to achieve desired electrical properties. Lithium-ion battery cathodes need exact stoichiometric ratios of lithium, cobalt, and oxygen for optimal energy density and cycle life.
2. Fundamental Concepts
The Mole Concept
The mole is the SI unit for amount of substance, defined as exactly 6.022 × 10²³ particles (Avogadro's number). This number was chosen so that one mole of carbon-12 atoms has a mass of exactly 12 grams, making atomic mass units (amu) directly convertible to grams per mole.
Why moles? Atoms and molecules are too small to count individually. The mole provides a bridge between the atomic scale (individual particles) and the laboratory scale (measurable masses). One mole of any substance contains the same number of particles, making stoichiometric calculations possible.
Example: One mole of water (H₂O) contains 6.022 × 10²³ water molecules. One mole of sodium chloride (NaCl) contains 6.022 × 10²³ formula units of NaCl.
Molar Mass
Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol). Calculate molar mass by summing the atomic masses of all atoms in the chemical formula.
Calculation Examples:
Water (H₂O):
2 hydrogen atoms: 2 × 1.008 g/mol = 2.016 g/mol
1 oxygen atom: 1 × 16.00 g/mol = 16.00 g/mol
Total molar mass: 18.02 g/mol
Glucose (C₆H₁₂O₆):
6 carbon atoms: 6 × 12.01 g/mol = 72.06 g/mol
12 hydrogen atoms: 12 × 1.008 g/mol = 12.10 g/mol
6 oxygen atoms: 6 × 16.00 g/mol = 96.00 g/mol
Total molar mass: 180.16 g/mol
Balanced Chemical Equations
A balanced equation has equal numbers of each type of atom on both sides, satisfying the law of conservation of mass. Balancing ensures that mole ratios accurately represent the reaction stoichiometry.
Balancing Steps:
Write the unbalanced equation with correct formulas
Count atoms of each element on both sides
Add coefficients (whole numbers in front of formulas) to balance
Check that all elements are balanced
Example - Combustion of Propane:
Unbalanced: C₃H₈ + O₂ → CO₂ + H₂O
Balanced: C₃H₈ + 5O₂ → 3CO₂ + 4H₂O
Check: Left side has 3 C, 8 H, 10 O. Right side has 3 C, 8 H, 10 O. Balanced.
Mole Ratios
Coefficients in balanced equations give mole ratios between any two substances. These ratios are the foundation of all stoichiometric calculations.
From C₃H₈ + 5O₂ → 3CO₂ + 4H₂O:
1 mole C₃H₈ : 5 moles O₂ (reactant to reactant)
1 mole C₃H₈ : 3 moles CO₂ (reactant to product)
5 moles O₂ : 4 moles H₂O (reactant to product)
3 moles CO₂ : 4 moles H₂O (product to product)
Use mole ratios as conversion factors. To find moles of CO₂ produced from 2 moles C₃H₈:
2 mol C₃H₈ × (3 mol CO₂ / 1 mol C₃H₈) = 6 mol CO₂
3. Step-by-Step Stoichiometry Calculations
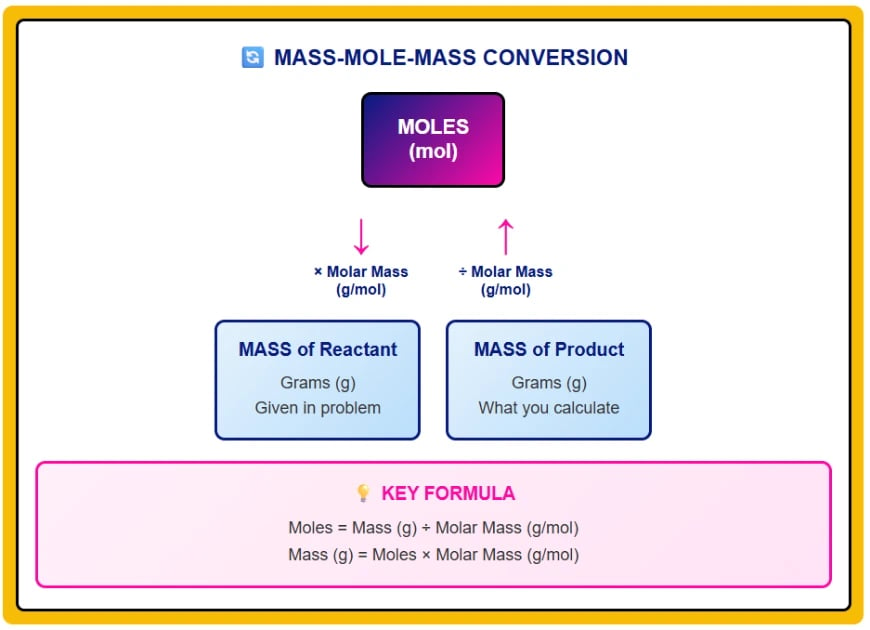
Method 1: Mass to Mass Conversions
The most common stoichiometry problem: given mass of reactant, find mass of product.
Step 1: Write Balanced Equation
Always start with a balanced equation. This provides the mole ratios needed for conversions.
Example Problem: How many grams of water are produced when 10.0 g of hydrogen reacts with excess oxygen?
Balanced equation: 2H₂ + O₂ → 2H₂O
Step 2: Convert to Moles
Convert given mass to moles using molar mass.
Molar mass of H₂ = 2.016 g/mol
Moles of H₂ = 10.0 g ÷ 2.016 g/mol = 4.96 mol H₂
Step 3: Use Mole Ratio
Apply mole ratio from balanced equation to find moles of desired substance.
From equation: 2 mol H₂ produces 2 mol H₂O (1:1 ratio)
Moles of H₂O = 4.96 mol H₂ × (2 mol H₂O / 2 mol H₂) = 4.96 mol H₂O
Step 4: Convert Back to Mass
Convert moles of product back to grams using its molar mass.
Molar mass of H₂O = 18.02 g/mol
Mass of H₂O = 4.96 mol × 18.02 g/mol = 89.4 g H₂O
Answer: 10.0 g of hydrogen produces 89.4 g of water.
Method 2: Mole to Mole Conversions
When given moles of one substance, find moles of another. This is the simplest stoichiometry calculation.
Example: In the reaction N₂ + 3H₂ → 2NH₃, how many moles of NH₃ form from 5.0 moles of N₂?
Use mole ratio directly:
5.0 mol N₂ × (2 mol NH₃ / 1 mol N₂) = 10.0 mol NH₃
Answer: 5.0 moles of nitrogen produce 10.0 moles of ammonia.
Method 3: Volume to Volume (Gases)
For gases at the same temperature and pressure, volume ratios equal mole ratios (Avogadro's Law). This simplifies stoichiometry for gas-phase reactions.
Example: What volume of oxygen is needed to react completely with 10.0 L of methane?
CH₄ + 2O₂ → CO₂ + 2H₂O
Volume ratio equals mole ratio:
10.0 L CH₄ × (2 L O₂ / 1 L CH₄) = 20.0 L O₂
Answer: 10.0 L of methane requires 20.0 L of oxygen (at same temperature and pressure).
4. Limiting Reactant Problems
What is a Limiting Reactant?
The limiting reactant is the reactant that is completely consumed first, stopping the reaction even if other reactants remain. It determines the maximum amount of product that can form. The reactant present in excess remains unreacted after the limiting reactant is consumed.
Analogy: Making sandwiches with 10 slices of bread and 3 slices of cheese. Each sandwich needs 2 bread slices and 1 cheese slice. You can make only 3 sandwiches before running out of cheese (limiting ingredient), leaving 4 bread slices unused (excess ingredient).
How to Identify Limiting Reactant
Method: Calculate how much product each reactant can produce. The reactant that produces the least product is the limiting reactant.
Example Problem: 25.0 g of aluminum reacts with 30.0 g of chlorine. Which is the limiting reactant?
2Al + 3Cl₂ → 2AlCl₃
Step 1: Convert both reactants to moles
Molar mass Al = 26.98 g/mol; Molar mass Cl₂ = 70.90 g/mol
Moles Al = 25.0 g ÷ 26.98 g/mol = 0.927 mol Al Moles Cl₂ = 30.0 g ÷ 70.90 g/mol = 0.423 mol Cl₂
Step 2: Calculate moles of product from each reactant
From Al: 0.927 mol Al × (2 mol AlCl₃ / 2 mol Al) = 0.927 mol AlCl₃ From Cl₂: 0.423 mol Cl₂ × (2 mol AlCl₃ / 3 mol Cl₂) = 0.282 mol AlCl₃
Step 3: Identify limiting reactant
Cl₂ produces less AlCl₃ (0.282 mol vs 0.927 mol), so Cl₂ is the limiting reactant.
Worked Example
Calculate the mass of AlCl₃ produced and the mass of excess Al remaining.
Mass of product: Molar mass AlCl₃ = 133.34 g/mol Mass AlCl₃ = 0.282 mol × 133.34 g/mol = 37.6 g AlCl₃
Mass of excess reactant: Moles Al consumed = 0.423 mol Cl₂ × (2 mol Al / 3 mol Cl₂) = 0.282 mol Al Moles Al remaining = 0.927 mol - 0.282 mol = 0.645 mol Al Mass Al remaining = 0.645 mol × 26.98 g/mol = 17.4 g Al
Answer: 37.6 g of AlCl₃ forms, with 17.4 g of aluminum left unreacted.
FAQ
What is stoichiometry in chemistry?
Stoichiometry is the quantitative study of reactants and products in chemical reactions, calculating the exact amounts of substances consumed and produced based on balanced chemical equations. The term comes from Greek words meaning "element" and "measure." Stoichiometry uses mole ratios from balanced equations to convert between masses, moles, and volumes of different substances. This allows chemists to predict how much product will form from given reactants, identify limiting reactants that determine maximum yield, and calculate reaction efficiency through percent yield. Stoichiometry is fundamental to chemical manufacturing, pharmaceutical synthesis, materials engineering, and environmental chemistry, ensuring processes use optimal reactant ratios to maximize product yield while minimizing waste.
How do you do stoichiometry calculations?
Follow a four-step method for stoichiometry calculations: (1) Write and balance the chemical equation to establish mole ratios between substances. (2) Convert the given quantity (usually mass in grams) to moles by dividing by molar mass. (3) Use the mole ratio from the balanced equation to convert moles of given substance to moles of desired substance. (4) Convert moles of desired substance back to the requested units (usually mass) by multiplying by molar mass. For example, to find grams of water produced from 10 g hydrogen: Balance equation (2H₂ + O₂ → 2H₂O), convert hydrogen to moles (10 g ÷ 2.016 g/mol = 4.96 mol), apply mole ratio (4.96 mol H₂ × 2 mol H₂O/2 mol H₂ = 4.96 mol H₂O), convert to grams (4.96 mol × 18.02 g/mol = 89.4 g H₂O). Always work through moles even when converting between masses.
What is a limiting reactant?
A limiting reactant is the reactant that is completely consumed first in a chemical reaction, determining the maximum amount of product that can form. Once the limiting reactant is used up, the reaction stops even if other reactants (excess reactants) remain. To identify the limiting reactant, calculate how much product each reactant can theoretically produce using stoichiometry. The reactant that produces the least amount of product is the limiting reactant. For example, if 25 g aluminum reacts with 30 g chlorine to form AlCl₃ (reaction: 2Al + 3Cl₂ → 2AlCl₃), convert both to moles, calculate product yield from each, and compare. Chlorine produces less product (0.282 mol AlCl₃ vs 0.927 mol), making it the limiting reactant. Understanding limiting reactants is crucial for maximizing product yield and minimizing waste in chemical processes.
How do you calculate percent yield?
Percent yield measures the efficiency of a chemical reaction by comparing actual yield (amount of product obtained experimentally) to theoretical yield (maximum amount predicted by stoichiometry). Calculate percent yield using the formula: Percent Yield = (Actual Yield / Theoretical Yield) × 100%. First, use stoichiometry to calculate theoretical yield from the limiting reactant. Then, measure the actual yield obtained from the experiment. Divide actual by theoretical and multiply by 100 to get percent yield. For example, if a reaction theoretically produces 127.1 g NaCl but only 115.0 g is recovered, percent yield = (115.0 / 127.1) × 100% = 90.5%. Percent yields less than 100% result from side reactions, incomplete reactions, product loss during purification, and measurement errors. Industrial processes optimize conditions to maximize percent yield, improving profitability and reducing waste.
What is the mole concept?
The mole is the SI unit for amount of substance, defined as exactly 6.022 × 10²³ particles (Avogadro's number). One mole of any substance contains this fixed number of atoms, molecules, ions, or formula units. The mole provides a bridge between the atomic scale (individual particles too small to count) and the laboratory scale (measurable masses). Avogadro's number was chosen so that one mole of carbon-12 atoms has a mass of exactly 12 grams, making atomic mass units (amu) directly convertible to grams per mole. For example, one mole of water contains 6.022 × 10²³ water molecules and has a mass of 18.02 grams (its molar mass). The mole concept is essential for stoichiometry because chemical equations show mole ratios between substances, allowing us to scale reactions from atomic proportions to practical laboratory or industrial quantities.
Why is balancing chemical equations important for stoichiometry?
Balancing chemical equations is essential for stoichiometry because the coefficients in balanced equations provide the mole ratios needed for all stoichiometric calculations. An unbalanced equation gives incorrect mole ratios, leading to wrong answers. For example, the unbalanced equation H₂ + O₂ → H₂O incorrectly suggests a 1:1:1 mole ratio. The balanced equation 2H₂ + O₂ → 2H₂O correctly shows that 2 moles of hydrogen react with 1 mole of oxygen to produce 2 moles of water. These mole ratios are the conversion factors used to calculate how much product forms from given reactants or how much of one reactant is needed to completely react with another. Balancing equations also satisfies the law of conservation of mass - atoms cannot be created or destroyed in chemical reactions, so the same number of each type of atom must appear on both sides of the equation. Without balanced equations, stoichiometric predictions are meaningless.
Related Articles
Continue exploring chemical calculations and materials science:
Ionic vs Covalent Bonds: Complete Guide - Understanding chemical bonding in reactions
Stratified vs Cluster Sampling: Data Science Guide - Statistical methods for materials research
Voice Biomarkers for Cognitive Decline Detection - Applications of chemical sensors in health tech
Sources and References
Stoichiometry - Wikipedia. Overview of quantitative relationships in chemical reactions. Wikipedia article
Mole (unit) - Wikipedia. Definition and applications of the mole concept. Wikipedia article
Chemical Equation - Wikipedia. Balancing equations and stoichiometric coefficients. Wikipedia article
Limiting Reagent - Wikipedia. Identification and calculations for limiting reactants. Wikipedia article
Chemistry: The Science in Context - Thomas R. Gilbert, Rein V. Kirss, Natalie Foster, Stacey Lowery Bretz. General chemistry textbook covering stoichiometry and mole concepts. Ung.edu
The Role of Industrial Chemistry in Modern Manufacturing - Applications of stoichiometry in chemical manufacturing and industrial processes. Omicsonline.org
